



Find all of your laboratory and workplace safety supplies at Safety Emporium!
 Poison |
 Glossary Index |
 PPE |
| MSDS Topics |
Free Sites | FAQ's | Regulations | Glossary | Software | Suppliers |
| Books | Forum | Poll | Fun stuff | Quiz | Store | |
| Understand your MSDS with the MS-Demystifier | Search ALL our MSDS info | |||||
Safety Emporium has all kinds of lab equipment such as stirring hot plates.
Examples of everyday monomers and their polymers include:
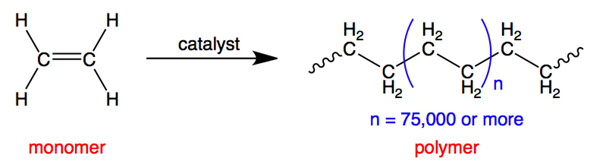
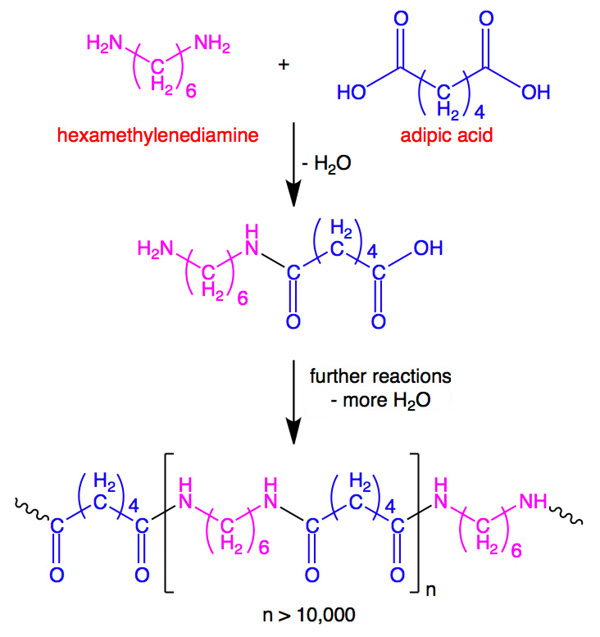

Get your laboratory equipment and glassware from Safety Emporium.
Polymerization of monomers is often an exothermic process (one that evolves heat). If polymerization begins when it is not desired, the result could be a fire or explosion. Materials that can spontaneously polymerize will usually list "hazardous polymerization" as a hazard in Section 10 (stability and reactivity) of their Safety Data Sheet.
Undesired polymerization of a monomer alters the physical properties of the material and could result in undesired changes. One fortunate example of this was the discovery of Teflon™ (the DuPont® trademark for poly(tetrafluoroethylene)) as a white solid in what was supposed to be a cylinder of gaseous tetrafluoroethylene. See the Further Reading below for more on this unlikely discovery.
See also: exothermic.
Additional definitions from Google and OneLook.
Entry last updated: Saturday, January 7, 2023. This page is copyright 2000-2025 by ILPI. Unauthorized duplication or posting on other web sites is expressly prohibited. Send suggestions, comments, and new entry desires (include the URL if applicable) to us by email.
Disclaimer: The information contained herein is believed to be true and accurate, however ILPI makes no guarantees concerning the veracity of any statement. Use of any information on this page is at the reader's own risk. ILPI strongly encourages the reader to consult the appropriate local, state and federal agencies concerning the matters discussed herein.