
|
The General Chemistry Demo Lab
Fun With Liquid Nitrogen
Introduction
Chemistry and temperature are intimately linked. As temperature is increased, molecules vibrate, move (translate) and rotate with increasing amplitude and energy. If enough energy is supplied to a material, chemical bonds can break and the material can change phase from a solid to a liquid to a gas or even a plasma. There is no practical limit on how high a temperature can get.
Likewise, as the temperature is reduced, the available energy for vibrating, translating and rotating atoms and molecules is also diminished. To be more accurate we should say this the other way around because molecular motion defines temperature! When we talk about an object's temperature, we are simply quantifying how much internal molecular/atomic motion it has.
If it were possible to stop all molecular motions, the temperature would be what scientists call absolute zero. Temperatures can never go lower than absolute zero; after all, how can one get fewer molecular motions than "none"?
Conventional temperature scales such as Celsius and Farhenheit were defined before such low temperatures were achievable. Zero on these scales was arbitrarily set and both scales are negative at a rather high temperature. In contrast, the Kelvin scale has no negative values - it sets zero to absolute zero (which is -459.67 ° F or -273.15 °C). The Kelvin scale is enormously useful to scientists and engineers, primarily because calculations are greatly simplified and standardized.
We have a handy online calculator that converts between the various scales, but that is not our focus here.
Let's look at the temperature range from absolute zero to the boiling point of water:
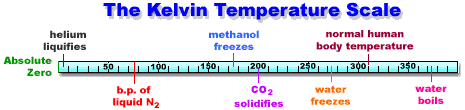
How easy is it to achieve absolute zero, 0 K? In a word, impossible. But scientists can get very close (down to milliKelvins or nanoKelvins) and some very unusual things start to happen such as Bose-Einstein condensations (the subject of the 2001 Nobel prize in physics). In a practical sense, around the laboratory, one can purchase liquid helium (b.p. = 3 K) although it is rather expensive.
The cheapest way of achieving relatively low temperatures in the laboratory is liquid nitrogen (b.p. = 77 K = -196 °C = -321 °F). Liquid nitrogen is readily available because it is prepared by liquifying air at 3,000 psi and then separating the nitrogen, oxygen and other important gases by fractional distillation. Because air is so abundant, liquid nitrogen costs only 20 to 25 cents per liter (not counting the expensive equipment you need to store it).
Liquid nitrogen is a tremendously useful cryogen (low temperature refrigerant) in the laboratory. It can be used to preserve tissue samples, collect gases, make scientific detectors work with greater efficiency, and to help achieve lower pressures in laboratory vacuum systems.
Of course, we have all know from science fiction movies that that liquid nitrogen has another important use. Killing Bad Guys. Simply spray a Bad Guy with liquid nitrogen for a few seconds to freeze him solid and simply hit, shoot, or drop him to shatter the Bad Guy into a billion tiny pieces...
In the Real World, it is not that simple. However, we can have a similarly enjoyable experience without the mess or those noisome capital murder charges by freezing and shattering smaller, everyday objects.
![]()
The Demo
There really isn't much to this demo. For maximum audience pleasure, we used an unsilvered Dewar flask so everyone can see the liquid nitrogen in action. A Dewar flask is essentially a Thermos - two layers of glass with a vacuum between them to provide superior insulation.
For this demo we are using a hemispherical Dewar that is housed in a home-made Lucite (Plexiglas) box. This arrangement is quite nice for demos - easy in/out for the items being frozen, great viewing, easy cleanup, and added safety in the event the Dewar implodes.
We were going to immerse some carnations or a rose (the classic demo) in this film clip, but none were available. Any object with a high water content works well, so we picked some fruit.
|
Click on the image at the right to view a short video of this demonstration. The freezing of the apple and banana takes several minutes, so we edited out everything but the fun parts. First, the objects are placed in the liquid nitrogen. This causes a great deal of nitrogen to boil off. The white fog you see is not the nitrogen gas (which is colorless), but water vapor condensing in the cold atmosphere above the liquid nitrogen. We hoped the apple would break when dropped on the table, but it "merely" became rock hard and bounced. It was then dropped on the floor. The banana was so brittle that it cracked while still in the Dewar! Of course, that did not interfere with our breaking and smashing. |
Note: a good scientist will perform the control experiment of smashing an unfrozen banana with a hammer. We leave that experiment up to the reader.
Other fun objects to freeze include squash balls (throw very hard against a nearby wall and they will shatter) and watermelons (just kidding).
![]()
Comments
Why does freezing a banana make it so brittle? Because bananas have a very high water content. When the banana was frozen, millions of tiny ice crystals formed. These crystals were not attached very well to one another because they each started growing at different spots and were not well-aligned. Scientists call crystal interfaces such as these grain boundaries.
The presence of many weak grain boundaries makes it very easy to shatter the banana. A good comparison is trying to break a large block of solid ice (one large crystal) versus breaking up a bag of ice cubes that are stuck together.
In addition, freezing the banana damages the cell walls and membranes, making them much weaker than normal. It is for reasons such as this that human cryonics - the idea of freezing a dead person and reanimating them in a few centuries, will likely remain science fiction for at least another several hundred years.
A discussion of why other materials get brittle (or harder) at low temperatures is beyond the scope of our discussion. Do a web search and you might find sites like this one which discusses why polymers get brittle in the cold.
Finally, we should note that we often see the the nitrogen/banana experiment described as using a frozen banana to drive a nail through a board. We've never seen that done successfully - the banana shatters too easily. Presumably, the frozen apple in our video clip would have been tough enough to do that.
![]()