|
Safety Emporium for all your lab and safety needs
 The Glassware Gallery
The Glassware Gallery
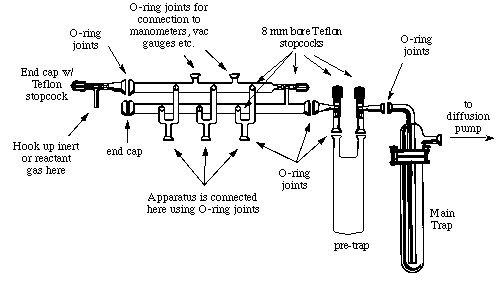
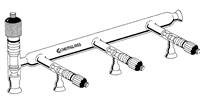
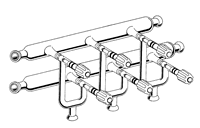
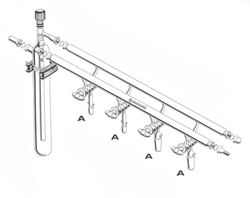
Schlenk Lines and Vacuum Lines
Introduction
One or two manifolds?
Safety Considerations
The use of high vacuum and Schlenk lines poses serious risks for injury or death. Some of the most important safety considerations include:- Explosion - Explosions can occur in a number of ways, including:
- The use of pressurized gases. High vacuum manifolds are often connected to an inert or reactant gas supply line. One must ensure that the vacuum system is not closed when the gas supply is opened - there MUST be a source of pressure relief such as a bubbler. The pressure must be monitored with an electronic gauge, manometer or bubbler; make sure the valve to the pressure reading device is open to the manifold!
Always TRIPLE CHECK that the manifold and supply line are connected to pressure relief (and your pressure sensor) before opening the gas supply. ALWAYS use an appropriate pressure regulator to avoid opening the line to more than 1 atm of pressure at any time.
- Condensed gases. Some gases, such as carbon monoxide and ethylene, are easily condensed into a liquid nitrogen-cooled trap. If the coolant level drops or you remove the nitrogen dewar without providing a means of pressure relief, the liquid may convert back to vapor. For example, just 10 mL of liquid CO (b.p. -191.5 °C) corresponds to 6.5 liters of gas. In a vacuum line with an internal volume of 500 ml the internal pressure would be 13 atm, more than enough to shatter the manifold with explosive force!
- Runaway reactions. Some reactions can occur violently and evolve large quantities of gas. Always provide a source of pressure relief!
- Heating a closed system. Never heat a vessel on a vacuum line without being open to a bubbler. Vacuum distillations must always have a pressure relief/regulator such as a manostat.
- Implosion - An unseen star crack or stress in a glass manifold can give rise to a catastrophic failure of the line while under pressure. Likewise, hitting the line with apparatus can cause a failure. While not usually as serious as an explosion, implosions generally involve sharp pieces of flying glass. And if the materials you are using are flammable or pyrophoric - watch out!
- Liquid oxygen - If a constant stream of air is pulled through a vacuum trap cooled with liquid nitrogen, liquid oxygen may condense in the trap (see item 1b above!). Liquid oxygen is exceedingly dangerous and reacts violently with most organic substances, including Teflon tape, vacuum grease, and organic solvents. Even without this consideration, the pressure generated when a small quantity of liquid oxygen vaporizes in a small space such as a vacuum manifold generates enough pressure to shatter the line.
Should you lower the trap on your line and find a pale blue liquid, immediately replace the trap and back away. Consult your supervisor IMMEDIATELY. Warn others of the danger, posting signs if necessary.
In addition, the use of vacuum pumps also poses a hazard should flammable liquids become entrained in the pump oil. See Brown, K. J., Smith III, A. B., Nguyen, M. "Lessons Learned from Laboratory Vacuum Pump Exhaust Explosion: Discussion and Preventative Strategies" ACS Chem. Health Saf. 2022, 29(1), pp 87-90 for an example of an explosion inside a vacuum pump cabinet during a rotary evaporation.
Disclaimer: This section discusses general dangers associated with high vacuum and Schlenk lines and is not meant to be a comprehensive reference to dealing with or assessing the hazards. There are many circumstances that have not been foreseen or discussed here and you should recognize the inherent danger in relying solely on this information!