



Find all of your laboratory and workplace safety supplies at Safety Emporium!
 Jaundice |
 Glossary Index |
 Ketosis |
| MSDS Topics |
Free Sites | FAQ's | Regulations | Glossary | Software | Suppliers |
| Books | Forum | Poll | Fun stuff | Quiz | Store | |
| Understand your MSDS with the MS-Demystifier | Search ALL our MSDS info | |||||
You can get cryogenic sublimators and other lab glassware from Safety Emporium.
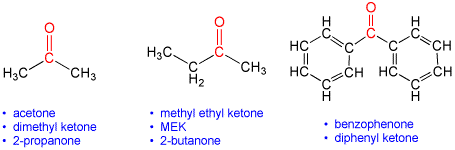
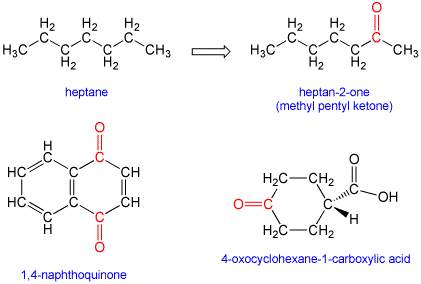
According to the International Union of Pure and Applied Chemistry, (IUPAC) naming (nomenclature) rules, simple ketones are named by taking the name of the longest acyclic hydrocarbon chain in the molecule, dropping the terminal "e" (if present), and adding the suffix "one" (pronounced with a one "o" as in "own). If the carbonyl is not in a terminal (end) position on the chain, then this is indicated by inserting a numeral before the suffix to indicate the position of the carbonyl carbon along the chain. For example, hept-2-one has the carbonyl group at the second carbon of the 7-carbon chain. In situations where there are other functional groups that take naming precedence, the ketone may be indicated by the use of "oxo". Certain other ketone-containing substructures have additional naming rules that are beyond the scope of our current discussion:
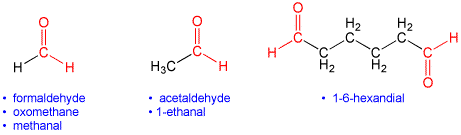
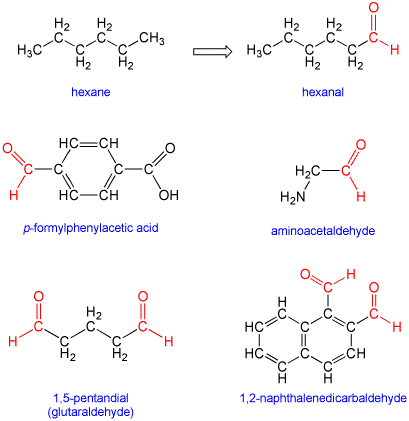
Under IUPAC nomenclature, aldehydes are named by taking the name of the longest acyclic hydrocarbon chain in the molecule, dropping the terminal "e" (if present), and adding the suffix "al", "aldehyde" or "carbaldehyde". In some cases the prefix "formyl" may be used. Two aldehydes are indicated by the suffix "dial". In addition, a number of trivial (traditional) names are still recognized. For detailed naming rules see Further Reading below. There is no need to use a number to indicate the position along the chain like there is with ketones because aldehydes are always terminal (end) groups.
Aldehydes and ketones are widely used industrial chemicals both as solvents and as chemical intermediates (ingredients for other chemicals). Most can be classified as volatile organic compounds meaning that their vapors may be easily inhaled or ignited. Many ketones and aldehydes are also flammable as liquids and solids.
Important note: formaldehyde is an industrially important aldehyde that is used on the billion ton scale. Glutaraldehyde is a "cold sterilent" used widely in the health care industry. Both are potent sensitizers. Exposure to either of these can make you hypersensitive to contact with other chemicals and carries definite health risks. Special rules and regulations have been developed to minimize formaldehyde exposure; see Further Reading below for more information.
Several ketones and aldehydes have been classified as known or likely carcinogens. For example, formaldehyde and acetaldehyde are known carcinogens. Studies are still underway on glutaraldehyde but it appears to have a much lower risk (if any) of carcinogenicity than formaldehyde. Acetone has not been reported to cause cancer.

Get your GHS-compliant labels and signs from Safety Emporium.
Ketones and aldehydes usually appear on Safety Data Sheets in one of two contexts:
Ketones may also be indirectly referenced in Section 11 (toxicological information) of the SDS, with the term ketosis, a medical condition in which ketones build up in the body.
The desirable chemical reactivity properties of aldehydes become undesirable when these chemicals enter your body, so be sure to read the label and SDS for any ketone or aldehyde-containing materials before use. You can find appropriate precautions to take when working with them on Section 8 (exposure controls/personal protection) of the SDS. Engineering controls such as fume hoods and personal protective equipment (PPE) such as gloves and respirators are a very good idea.

Get your pure water valves and faucets from Safety Emporium.
See also: alcohol, carboxylic acid, chemical formula, carbon monoxide, ketosis, VOC.
Additional definitions from Google and OneLook.
Entry last updated: Monday, January 2, 2023. This page is copyright 2000-2025 by ILPI. Unauthorized duplication or posting on other web sites is expressly prohibited. Send suggestions, comments, and new entry desires (include the URL if applicable) to us by email.
Disclaimer: The information contained herein is believed to be true and accurate, however ILPI makes no guarantees concerning the veracity of any statement. Use of any information on this page is at the reader's own risk. ILPI strongly encourages the reader to consult the appropriate local, state and federal agencies concerning the matters discussed herein.