 |
 Safety Emporium is a proud sponsor of this site. |
 |
Home Page --> Scientific Glassblowing Demonstrations
|
 |
 Safety Emporium is a proud sponsor of this site. |
 |
Home Page --> Scientific Glassblowing Demonstrations
|
Effective teaching requires clear communication and the creation of a comfortable, friendly environment or atmosphere in which to learn. Demonstrations that allow audience participation tend to be the most successful and the educational content can be specifically tailored to your audiences' age and capabilities.
The content of these demonstrations is inherently scientific. The intent is to promote the sciences in general and scientific glassblowing in particular. When possible, try to coordinate the glassblowing with concurrent student projects. For example, an experiment in the chemistry lab may mean more to a student if he/she uses a test tube that he/she has made.
Safety notes - Safety is always our first concern when working with glass, flames, and compressed gases. Set up your demonstration area in a manner to protect people and property. Use appropriate protective clothing and safety glasses, comply with all safety and fire regulations and guidelines and work with an assistant. |
Good demonstrations involve participation:
Note: To encourage student participation, I have added some peer-led leading to my most recent demonstrations. Selected students spend a half day at the glassblowing facility learning some basic glassblowing skills and learning more about the glassblowing shop. Those students then become the "instructors" to their classmates when we do the demonstration a day or so later. The student instructors do an admirable job of sharing information with the class, answering questions and encouraging participation.
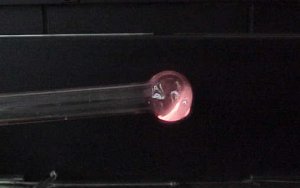
This demonstration allows students of all ages to participate without handling hot glass or the torch, and without coming close to the flame. The key is to have a rather long blowhose (with individual throw-away mouthpieces), keeping the students away from the hot glass and flame, but still close enough to observe the glass reacting to their "blowing" into the blowhose.
Some suggestions and questions to ask during an effective demonstration include:
 |
 |
 |
 |
 |
 |
This demonstration simply shows various methods of "breaking" glass, but in a way that we want it to break. Some suggestions/approaches here include:
This demonstration requires more preparation than most, but is a good way to incorporate a little "chemistry" into the lesson plan. This demonstration has been successfully given to students in the 6-8 year old bracket, but may be better suited for those a little older. The amount of technical information given may vary quite a bit depending on the age group.
For the glass preparation you can have the students blow bulbs (using the same basic instruction as in Blowing Bubbles). 19 mm medium wall tubing works well. Try to make bulbs about 25-30mm as your objective and be sure they are not thin!
Tips for the silvering:
This demonstration requires assistance and works well in breaking up the class into two groups, maintaining interest levels over an extended period of time.
A few minutes of your time can create lifetimes of happy memories. Here are just a few examples of the rewards you can reap with effective glassblowing demonstrations:
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
Disclaimer: Glassblowing has inherent hazards that can result in burns, cuts, injuries or even death. While every effort has been made to convey best safety practices, the materials presented herein can not anticipate every possible risk or hazard associated with a given glassblowing activity. The reader is responsibile for assessing and mitigating any and all such hazards or risks before commencing any activity connected with his or her use of these materials. Neither the author nor ILPI assumes responsibility or liability for injuries, losses, claims or damages to the reader or other parties that may result from the use or misuse of these materials.
by Joe Walas and/or ILPI, all rights reserved unless otherwise stated. Unauthorized duplication or posting on other web sites is expressly prohibited. Send suggestions and comments (include the URL if applicable) to us by email.